💧 Product Description
Our purified water systems integrate GMP-compliant design with multi-stage purification: pre-filtration, RO membranes, and EDI modules ensure water quality meeting USP <643> (conductivity ≤1.3μS/cm, TOC ≤500ppb). Constructed from passivated 316L stainless steel (Ra≤0.8μm), the systems feature sanitary welds and dead-leg-free piping (DN/OD≤3:1) for bioburden control. Automated Pasteurization / Ozone cycles validate disinfection efficacy, while PLC-controlled recirculation (≥1.5m/s) maintains continuous purity. Engineered for seamless IQ/OQ/PQ validation, the systems optimize regulatory compliance and operational reliability in pharmaceutical manufacturing.
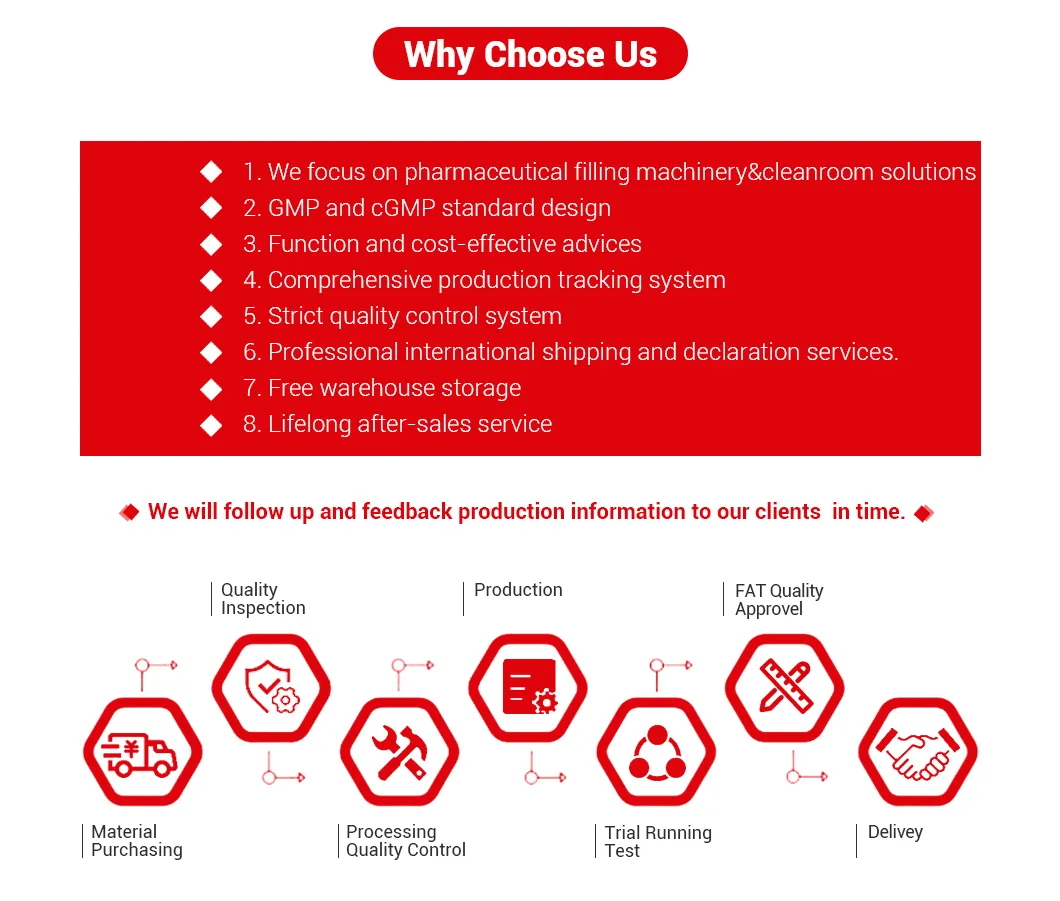
🌟 Key Features and Advantages
01. Modular design, compact structure, easy and fast field installation.
02. Advanced automatic control mode, stable and reliable operation.
03. Double softeners in series or parallel for effective microbial control.
04. Full pasteurization and flexible disinfection methods.
05. Unique circulation mode to prevent bacteria breeding.
06. Constant pressure control to prolong EDI service life.
07. Automatic adjustment of reverse osmosis recovery for energy saving.
08. Flexible water supply to reduce shutdown time and improve equipment life.
❓ Frequently Asked Questions
What water quality standards do these systems meet?
Our systems are designed to meet USP <643> requirements, ensuring conductivity ≤1.3μS/cm and TOC ≤500ppb, which are standard for pharmaceutical and industrial purified water.
What materials are used for the piping and components?
All water-contact parts are constructed from passivated 316L stainless steel with a surface roughness of Ra≤0.8μm to prevent contamination and ensure sanitary operation.
How does the system prevent bacterial growth?
We utilize automated pasteurization, ozone cycles, and a continuous circulation mode (recirculation speed ≥1.5m/s) to eliminate dead legs and control bioburden effectively.
Is the system difficult to install on-site?
No, the system features a modular design and compact structure, which allows for fast and easy field installation, minimizing downtime at your facility.
How does the system optimize energy consumption?
The reverse osmosis recovery rate can be adjusted automatically, and the system can be equipped with a concentrated water recovery device to significantly save energy and water.
What kind of control system is provided?
The systems are equipped with advanced PLC-controlled automation, providing stable, reliable operation and simplified management of the purification process.